Balance of inputs to the striatum shifted towards the direct-pathway neurons in vivo, but not in Parkinson’s disease
Parkinson’s disease (PD) is a neurodegenerative disease in which neurons that produce the neuromodulator dopamine progressively die. Loss of dopamine results in debilitating motor and cognitive symptoms. Over the last decade, researchers identified a number of changes that occur in the brain during Parkinson’s disease, but some very basic information is still missing.
“Even though we know a lot about the genetic, chemical, morphological and electrophysiological changes associated with PD, it is quite frustrating that we still do not know much about cortical inputs into the striatum”, says Arvind Kumar. The striatum is the main input station of the basal ganglia, and is responsible for tasks such as motor control and reward learning. Within the striatum, two main groups of cells orchestrate the proper functioning of the basal ganglia through the so-called “direct” and “indirect pathways”. Correspondingly, these cells are called direct-pathway and indirect-pathway medium spiny neurons (dMSNs and iMSNs for short). “To understand the symptoms of Parkinson’s disease we need to find out how the connectivity of dMSNs and iMSNs is altered during PD”, explains Marko Filipović, one of the two lead authors of the new study.
Previously, the group of Arvind Kumar and Prof. Dr. Ad Aertsen at BCF used computational models to estimate the strength of inputs to the dMSNs and iMSNs. Already in 2015 they predicted that dMSNs should receive more input than iMSNs (Bahuguna et al. 2015). “While some in vitro studies provided support for our theoretical prediction, we lacked in vivo measurements to critically test it” says Ad Aertsen. But that is easier said than done. “To verify the theoretical predictions we need to simultaneously record activity of a pair of connected neurons in the neocortex and in the striatum – an extremely daunting task”, adds Gilad Silberberg, a leading experimental neuroscientists based at Karolinska Institute, Stockholm, Sweden.
As part of the answer to this problem, Maya Ketzef and Ramon Reig, two researchers from Gilad Silberberg’s group, had previously recorded in vivo membrane potentials of dMSNs and iMSNs from healthy and dopamine-depleted anesthetized mice (Ketzef et al. 2017). Filipović and Ketzef then analyzed the fluctuations and the power spectra of the recorded potentials, applying principles of linear systems theory that suggested that the size of the membrane potential fluctuations is proportional to the amount of synaptic input. The differences they found in the membrane potential spectra for the first time provided evidence that in healthy animals in vivo dMSNs indeed receive stronger input than iMSNs, as predicted by the computational models. More importantly, they found that this difference in inputs of the dMSNs and iMSNs is lost in an animal model of Parkinson’s disease.
These results highlight the importance of computational models in neuroscience. “Without the computational model we would neither have posed the question whether there are differences in the inputs to the two types of neurons, nor whether we could find the answer by analyzing the already available membrane potential data”, says Arvind Kumar.
While these results are very promising, we still do not know the exact nature of the inputs to the two types of neurons. Gilad Silberberg sums up: “With the methods used we cannot determine if the difference we see is because dMSNs receive more excitatory or more inhibitory input.” Indeed, new experiments are needed for better characterizing the synaptic inputs to dMSNs and iMSNs. In any case, this study shows that an appropriate combination of theory and computational modelling can help extract more information from experimental data.
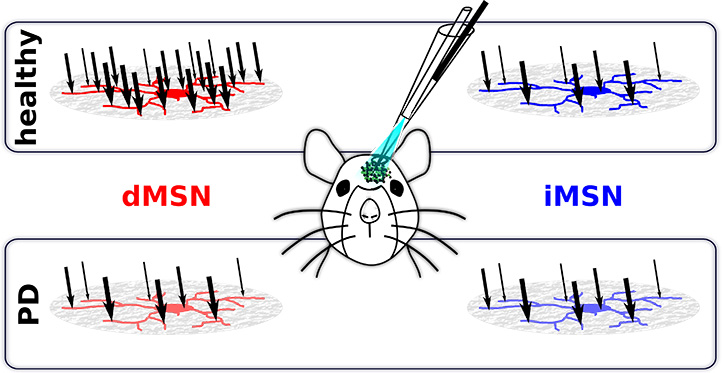
Figure Caption
Using recordings of in vivo membrane potentials of striatal medium spiny neurons in anesthetized mice, researchers found that synaptic input (black arrows) to dMSNs (red cells) is stronger than to iMSNs (blue cells). This difference is lost in Parkinson's disease.
Original Publication
Filipović M, Ketzef M, Reig R, Aertsen A, Silberberg G, Kumar A (2019) Direct pathway neurons in mouse dorsolateral striatum in vivo receive stronger synaptic input than indirect pathway neurons. J. Neurophysiology (ahead of print)
Further Publications
Bahuguna J, Aertsen A, Kumar A (2015) Existence and control of Go/No-Go decision transition threshold in the striatum. PLoS Computational Biology 11(4):e1004233.
See also
To Go or Not to Go
Ketzef M, Spigolon G, Johansson Y, Bonito-Oliva A, Fisone G, Silberberg G (2017) Dopamine depletion impairs bilateral sensory processing in the striatum in a pathway-dependent manner. Neuron. 94(4):855-65.
Contact
Marko Filipović
+49 (0)761 203 9324
E-mail: marko.filipovic@bcf.uni-freiburg.de
Prof. em. Dr. Ad Aertsen
E-mail: ad.aertsen@biologie.uni-freiburg.de
University of Freiburg
Bernstein Center Freiburg
Hansastr. 9a
79104 Freiburg
Germany